MODAL / RAYON / LYOCELL
WE ARE SPECIALIZED
RAYON FIBERS
Rayon is derived from French word “rays of light” and was first sold as artificial silk.
Rayon is described as a regenerated fiber because the cellulose, obtained from softwoods or from the short fibers (linters) that adhere to cottonseeds, is converted to a liquid compound, squeezed through tiny holes in a device called a spinneret, and then converted back to cellulose in the form of fiber.
The history of cellulose fibers dates back to the 1860s when the first rayon fibers were commercialized by Courtaulds. But the so-called rayon process includes toxic chemical treatments to block hydroxyl groups of cellulose to prepare a spinnable solution, sometimes causing ecological problems. The first practical steps toward producing such a fiber were represented by attempts to work with the highly flammable compound nitrocellulose, produced by treating cotton cellulose with the nitric acid.
In 1884 and 1885 in London, British chemist Sir Joseph Wilson Swan exhibited fibers made of nitrocellulose that had been treated with chemicals in order to change the material back to nonflammable cellulose. Swan did not follow up the demonstrations of his invention; thus, the development of rayon as a practical fiber really began in France, with the work of industrial chemist Hilaire Bernigaud, Comte de Chardonnet
who is frequently called the father of the rayon industry. In 1889 Chardonnet exhibited fibers made by squeezing a nitrocellulose solution through spinnerettes, hardening the emerging jets in the warm air, and then reconverting them to cellulose by chemical treatment. Manufacture of “Chardonnet silk,” an early type of rayon and the first commercially produced man-made fiber, began in 1891 at a factory in Besançon.
Although Chardonnet’s process was simple and involved a minimum of waste, it was slow, expensive, and potentially dangerous. In 1890 another French chemist, Louis-Henri Despeissis, patented a process for making fibers from cuprammonium rayon. This material was based on the Swiss chemist Matthias Eduard Schweizer’s discovery in 1857 that cellulose could be dissolved in a solution of copper salts and ammonia and, after extrusion, be regenerated in a coagulating bath. In 1908 the German textile firm J.-P. Bemberg began to produce cuprammonium rayon as Bemberg silk.
The third type of cellulose and the type most commonly made today was produced in 1891 from a syrupy, yellow, sulfurous-smelling liquid that three British chemists Charles F. Cross, Edward J. Bevan, and Clayton Beadle discovered by dissolving cellulose xanthate in dilute sodium hydroxide. By 1905 the British silk firm Samuel Courtauld & Company was producing this fiber, which became known as viscose rayon (or simply viscose). In 1911 the American Viscose Corporation began production in the United States.
It is interesting to note that the first rayon factory in India was started in the year 1951. At present, there are eleven major rayon factories in India. Rayon is a major feedstock in the manufacture of carbon fiber. Grasim of India is the major producer of rayon in the world which is claiming 24% market share. Trade names are used within the rayon industry to label the type of rayon in the product. Viscose Rayon was first produced in Coventry England in 1905 by Courtaulds. Grasim of India is the largest producer of rayon in the world (claiming 24% market share). It has plants in Nagda, Kharach, and Harihar – all in India, as well as joint ventures in Canada, Laos, and China.
According was a major manufacturer of cellulose-based fibers and yarns. Production facilities can be found throughout Europe, the U.S., and Brazil. North American Rayon Corporation of Tennessee produced viscose rayon until its closure in the year 2000.
Cellulose is a chief structural component to which plants owe their rigidity and form. Cellulose is a natural polymeric material having a fibrous structure. It is the principal component of the covering of plant cells. Cellulose represented by the formula (C6H10O5) is a linear polymer containing glucose units. Wood, cotton and other plants material are the main sources of cellulose. In the pure form, cellulose is present in cotton (98%) while in wood it is about 50% of the dry wood. Some of the natural materials based are casein fiber, alginate fiber, vicar, soyabin fiber amongst the various rayon viscose rayon is most commonly commercially manufactured rayon and find wide application in textile, tire cord industry.
Because rayon is manufactured from naturally occurring polymers, it is considered a semi-synthetic fiber. Specific types of rayon fibers are:
- Chardonnet silk / First Rayon fibers (discovered in 1885)
- Cuprammonium Rayon or Bemberg Rayon (discovered in 1891)
- Viscose Rayon (discovered in 1905)
- Acetate Rayon
- Lyocel (Trade name Tencel ) (discovered in 1972)
- Bamboo Rayon (discovered in 2003)
- Pyroxylin rayon
- Modal
Bemberg is a trade name for cuprammonium rayon developed by J. P. Bemberg. Bemberg performs much like viscose but has a smaller diameter and comes closest to silk in feel. Bemberg is now only produced in Italy due to the United States Environmental Protection Agency regulations in the US. The fibers are finer than viscose rayon.
Modal and Tencel are widely used forms of rayon produced by Lenzing AG. Tencel, generic name lyocell, is made by a slightly different solvent recovery process and is considered a different fiber by the US FTC. Tencel and lyocell were first produced commercially by Courtaulds’ Grimsby plant in England. The process, which dissolves cellulose without a chemical reaction, was developed by Courtaulds Research.
Lyocell rayon is a flame retardant form of viscose which has silica embedded in the fiber during manufacturing.
Out of these rayon’s, viscose rayon is the most important from the viewpoint of quantity. More than 80 % of the world’s production of filament rayon is of Viscose only. Next in order comes acetate with about 16% and Cuprammonium with about 4%.
The four methods of manufacturing rayon are the
Nitrocellulose processes
Viscose process
Cuprammonium process and
Process for making saponified rayon.
Some Characteristics of rayon fiber highly absorbent, Soft and comfortable, Easy to dye and drape well. Such fibers are designated as high tenacity rayons, which have about twice the strength and two-thirds of the stretch of regular rayon. An intermediate grade, known as medium tenacity rayon, is also made. Its strength and stretch characteristics fall midway between those of high tenacity and regular rayon
CUPRAMMONIUM RAYON
The fabric is commonly known by the trade name “Bemberg”, owned by the J.P. Bemberg company. who improved it and first produced commercially in 1901. The fabric may also be known as “cupro” or “
Cuprammonium rayon is simply cellulose dissolved in a cuprammonium solution. The cuprammonium process converts cellulose to a soluble compound by combining it with copper and ammonia 12. It was originally called artificial silk or wood silk. Rayon is a regenerated fiber because cellulose is converted to a liquid compound and then back to cellulose in the form of fiber for ex: cuprammonium rayon is made by dissolving cellulose is an ammoniacal copper sulfate solution
Cuprammonium rayon is produced by a solution of the cellulosic material in cuprammonium hydroxide solution at the low temperature in a nitrogen atmosphere, followed by extruding through a spinneret into a sulphuric acid solution necessary to decompose cuprammonium complex to cellulose. This is a more expensive process than that of viscose rayon. Its fiber cross-section is almost round.
Manufacture of Cuprammonium Rayon: The source of cellulose for this rayon is cotton
- Mechanical Treatment
- Chemical Treatment
- Mechanical Treatment: The cotton liners are transported in bales in highly compressed state and the object of the mechanical treatment is to loosen them and to remove mechanically admixed and loosely bound impurities such as dust sand, seed residues etc.
- Chemical Treatment: The mechanically opened and purified cotton
linters are boiled under pressure for several hours with dilute sod ash ( Na2Co3) solution (2%) to which a little amount of caustic soda may be added. The natural fatty matter present in the cotton is converted into soluble substance by the action of soda ash and thus removed from cottonlinters .
Materials and Methods
Preparation of cuprammonium biodegradable rayon threads from different types of paper material such as Whatman filter paper, ordinary filter paper and A4 size printable papers involved following.
A. Preparation of Schweitzer’s Solution
Copper
B. Preparation of Cellulose material
2g of paper materials cut into small pieces and transferred to the conical flask to this tetra-ammine cupric hydroxide solution was added. The flask was capped and kept for 15 days, during this period the paper is dissolved completely.
C. Formation of Rayon Thread
Distilled water (50ml) was taken in the beaker, so this 20ml
CuSO4 + 2NH4OH Cu(OH)2 + (NH4 )2SO4
Cu(OH)2 + 4NH4OH [Cu(NH3 )4](OH)2 + 4H2O>
[Cu(NH3 )4 ](OH)2 + Pieces of filter paper for 10-15 days give a viscous solution called viscous
Properties of Cuprammonium Rayon The one important characteristic of these fibers is their extreme fineness. Filaments as fine as 1.33 deniers are produced regularly ( as compared to viscose rayon which has a usual denier of around 2.5). This increased fineness is due to the stretch that is applied to the filaments during spinning.
- Because of its fineness, cuprammonium rayons produce a soft silk-like a handle.
- It has all the properties of cotton except that the average DP is lower and a larger portion of this fiber is occupied by amorphous regions. Hence the rayon swells to a greater extent and hence chemical reactions take place faster in the case of rayon than in the case of cotton.
- Like viscose rayon, it burns rapidly and chars at 180 deg C. It is degraded and weakened by exposure to sunlight in the presence of oxygen and moisture. On ignition, it leaves behind ash containing copper.
- The average tensile strength of cuprammonium rayon is 1.7-2.3 in dry and 0.9-2.5 in a wet state.
- It has an elongation at break of 10-17% when dry.
- Moisture content at 70 deg F and 65% RH is about 11% as in case of Viscose Rayon.
- Dye absorption power for direct dyes of cuprammonium rayon is greater and shades obtained are deeper than viscose rayon.
- The filaments appear uniform with surfaces having no markings, in the longitudinal view. Cross sections are round and smooth with occasionally slightly oval.
Viscose Rayon
The name Viscose was derived from the word “vicious” which means sticky spinning solution out of which “Rayon” was manufactured. Thus the innovative cellulosic derivative has taken the present name of “Viscose rayon”.
The name rayon referred to as a generic term desired to replace the word ‘artificial silk’. It is the most versatile and the most widely used manmade fiber. It is the cheapest fiber because it is made from cellulose extracted from wood pulp, which is cheap and widely available raw material produced in areas unsuitable for food production. It is produced in a wide range of filament thickness ranging from the fineness of natural silk to the thickness of coarse wool or hair.
Various types of viscose rayon are: High tenacity rayon
- High wet modulus (HWM) rayon,
- Flame retardant rayon
- super adsorbent rayon
Rayon is the oldest commercial manmade fiber.
Rayon fiber was prepared from different paper involved converting cellulose into a liquid compound. Viscose fiber rayon was obtained, when ammonia reacted with copper
The process used to make viscose can either be a continuous or batch process. The batch process is flexible in producing a wide variety of rayons having broad versatility. Rayon’s versatility is the result of the fiber being chemically and structurally engineered by making use of the properties of cellulose from which it is made. However, it is somewhat difficult to control uniformity between batches and it also requires high labor involvement. The continuous process is the main method for producing rayon. The solubility test was performed for identification of rayon.
The methods mentioned, the viscose method is relatively inexpensive and of particular significance in the production of nonwoven fabrics. Some Characteristics of rayon fiber highly absorbent, Soft and comfortable, Easy to dye and drape well. Such fibers are designated as high tenacity rayons, which have about twice the strength and two-thirds of the stretch of regular rayon. An intermediate grade, known as medium tenacity rayon, is also made. Its strength and stretch characteristics fall midway between those of high tenacity and regular rayon
- Raw Material and its preparation: The raw material for the production of viscose rayon is wood pulp. There are many types of wood from which we can get this wood pulp but for the viscose rayon, we consider only those which contain more than 45% α-cellulose. α-cellulose is one which has much greater D.P. hen 1000 while β – cellulose and γ – cellulose have less D.P. than 900. Cotton linens (small cotton fibers 2-5 mm length) contain 98 % cellulose with a D.P. of 10000 to 20000. But they are used for proper industry and not available for viscose rayon production.
The different types of plant are containing such α-cellulose:- Eucalyptus plants ——————– 60 to 70 % α-cellulose.
- Bamboo plants ———————– 45 to 50 % α-cellulose
- Oaktree also contains α-cellulose.
But the principal source of cellulose used for viscose rayon manufacturer is wood pulp obtained from the tree, (especially spruce tree) pulp that has been bleached to freet from gums, lignin, and other impurities. The pulp is received in the factory in the form of boards or sheets of about 52 cm square x 0.08 cm thick i.e. 24” x 18” x ½”. These sheets are conditioned to absorb the standard moisture of about 6 % that is relevant for the processing.
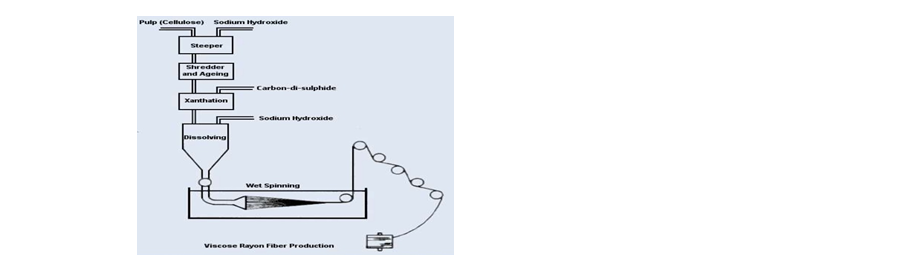
- Steeping: It involves the treatment of the pulp boards with caustic soda solution of mercerizing concentration (18% NaOH) to form the alkali cellulose.Cell – OH + NaOH Cell-O-Na +H2O
Soda Cellulose or Alkali Cellulose
(C6H10O5)n + n NaOH (C6H9O4Na)n + nH2O
This process is known as steeping or mercerizing. The board becomes greatly swollen.
 Actually, the operation carried out in a long rectangular tank in which a hydraulic ram is incorporated. Thus, when mercerization is complete, the excess alkali is pressed out by a hydraulic ram in the press – leaving the softened boards of alkali – cellulose containing known quantities of caustic soda water. This takes nearly 14 hours. The `dark brown colored caustic soda solution is then drained off and the sheets pressed by the movable plates to squeeze the caustic soda solution to a given moist mass of soda cellulose, which is then transferred to a shredding machine for the next operation.
Actually, the operation carried out in a long rectangular tank in which a hydraulic ram is incorporated. Thus, when mercerization is complete, the excess alkali is pressed out by a hydraulic ram in the press – leaving the softened boards of alkali – cellulose containing known quantities of caustic soda water. This takes nearly 14 hours. The `dark brown colored caustic soda solution is then drained off and the sheets pressed by the movable plates to squeeze the caustic soda solution to a given moist mass of soda cellulose, which is then transferred to a shredding machine for the next operation.
- Shredding: To increase the surface of the alkali cellulose so as to ensure rapid and uniform processing at subsequent stages, the alkali cellulose is now mechanically disintegrated or shredded.
The shredding machine consists of a drum inside which revolve a pair of blades with serrated edges. The shredders are water cooled and will take 200 lbs of pressed soda – cellulose at a time. In 2 – 3 hours they break it up into fine ‘crumbs’ resembling breadcrumb. - Aging: Ageing consists essentially of storage under controlled atmospheric conditions, oxidation and depolymerization occur and these changes considerably influence the properties of the viscose yarn that will eventually be made. (Depolymerization (or depolymerization) is the process of converting a polymer into a monomer or a mixture of monomers. All polymers depolymerize at high temperatures, a process driven by an increase in entropy.) The crumbs of soda – cellulose are stored in small galvanized steel drums for about 48 hours (1 to 3 days) at 230C. The time and temperature depend on the nature of the pulp, the degree of aging (maturity) desired, spinning process to be employed etc. 5. Xanthation / (Churning):

This process is also named as subsidizing. After the aging process, the crumbs of soda cellulose are transferred to rotating airtight churners (water jacketed hexagonal churner). These are double jacketed so as to maintain the required temperature during churning. Carbon disulfide is slowly admitted as a gas or liquid through a perforated pipe within the drum the quantity of CS2 being 32 to 35 % of the weight of cellulose in alkali cellulose. The reaction is permitted to continue for 1 to 3 hours, the churn being rotated at a slow speed (1 to 4 rpm), colors of product changes from white to yellow to deep yellow and then to reddish orange or called carrot color. Sodium cellulose xanthate formed during this process should not be allowed to discover in the subsequent process. After the churning is over vacuum is applied for removing the excess of CS2 from the mixer.
- Mixing and Dissolving : At this stage the sodium cellulose xanthate, an orange-colored product is to a cylindrical vessel equipped with agitators and dilute solution of caustic soda (18% NaOH) in calculated quantity is added so that the sodium cellulose xanthate gets dissolved, the dissolving being carried out by stirring the contents for 4 to 5 hours at a temp. of 20 to 26 0C (Cooling).In this way, the sodium cellulose xanthate to give a clear brown, the viscous liquid similar in appearance to honey but still it is impure too aerated and too young to be spun. This solution is blended in big mixers to homogenize any irregularity and to get more uniform characteristics in the final product. If a delustred yarn is eventually required, a pigment, perhaps 2 % on the weight of the cellulose of TiO2 is added to this solution and dispersed by stirring.
- Filtration / De-aeration: This is necessary before the spinning stage is reached since the rayon is to be formed by forcing the solution through the spinneret having very small holes, all bubbles and solid particles likely to choke the spinneret holes must be removed. Hence the viscous solution is passed through a series of filter presses on its way to the large tanks in which it is ripened and later the solution is again filtered several times in the same manner while being transferred from one container to another. Finally, de-aeration is accomplished i.e. air bubbles are removed by maintaining a partial vacuum over the viscose solution in the airtight tanks in which it is kept during the last part of the ripening periods.
- Ripening: As the alkali – cellulose needs
ageing this viscous solution (Viscous Solution) requires to ripen to give a solution having the best spinning qualities. The stirring being stopped now and the viscose is stored for 4 to 5 days at a controlled constant temperature of 10 to18 0C.
During this period, the viscosity at first falls and then rises to the original valve. Again all the air bubbles are removed by De-aeration process by exposure to a vacuum and filtered carefully. The solution is now ready for spinning to produce viscose rayon filaments by the wet spinning method. - Extrusion or Spinning: The spinning of viscose into a fiber involves the basic reaction of regeneration and is simply the recovery of cellulose from cellulose xanthate by acid decomposition reaction upon entering the bath, the viscose is coagulated and acid diffuses into the filament regenerating cellulose.
During this time, viscose fiber is also stretched to orient the cellulose molecules in the direction of the fiber axis.
Metered pumps force a measured amount of solution through the multi-holed spinneret, which is arranged in a row, in a long tank, horizontally into a coagulating bath.
Spinneret is designed in a variety of manner to cater to the requirements of wet, dry and melt spinning. It is a plate which is precision bored with orifices of diameter. Usually between 0.05 to0.01 mm. ( the size of the holes can be varied according to the requirements. The spinneret for filament yarn production has few tens or hundreds of holes whereas for staple fiber production have few hundreds or thousands of holes. The shape of orifices determines the cross-section of the fibers.
Denier of filaments is controlled by the number of holes in the spinneret as well as the stretch ratio.
The spinneret in case of wet spinning of viscose rayon is submerged in a coagulating bath containing:
- 10 % H2 SO4 (Regenerating)
- 18% Na2 SO4 (Coagulant)
- 1% Zn So4 (Added strength)
The bath temperature is maintained at 40 to 45 0C sodium sulfate acts as a coagulant and precipitates or coagulates the dissolved sodium cellulose xanthate and H2 SO4 converts the xanthate into cellulose (regeneration process) carbon disulfide and sodium sulfate.

The glucose is supposed to give softness and pliability to the filaments while Zn SO4 gives added strength
- The quality of viscose rayon this formed depends on:
- The temperature of the spinning bath
- The composition of the spinning bath
- The speed of precipitation of the filaments
- The period of immersion of filaments in the spinning bath
- The speed of the spinning
- The stretch imparted to the filaments
A number of separate filaments are continuously formed and they are steadily drawn away from the spinneret under a moderate degree of tension and they led to an eye (guide) and is collected while being given a stretch by means of paid guide rollers and given a small degree of twist in a rapidly rotating spinning pot named as Topham Box.
Topham box is the take-up the device in the form of a bucket on a spindle rotating at a speed of 6000 to 7000 rpm. About 2 lbs of standard viscose can be cake spun and packed in the Topham box. After Topham box is completed then it is replaced by the another. This cake may be converted into the hank form with the help of the reeling process.
The yarn collected in this way is impure and relatively weak which is to be purified. Purification consists of four operations as below:
- Washing
- Desulphurising
- Bleaching
- Washing
Physical Properties of Viscose Rayon:
Like polyester or other manmade fiber, viscose rayon also consists of some important physical properties, important physical properties of viscose rayon fiber as below :

PHYSICAL PROPERTIES
- Moisture Absorption: Absorbency relies on the inner structure of regenerated, cellulose, which is highly amorphous. It can be penetrated readily by water and other aqueous solutions. Due to its absorbency, it can be readily dyed and it absorbs perspiration easily. As water enters the fiber, it makes the hydrogen bonds between the molecular chains, push them apart and causes the fiber to swell. In this swollen state, the fibers are weaker. Garments made of rayon shrink easily and clothes dry steadily. It absorbs more moisture than cotton. Moisture Content of Coton is 6% at 70 deg F and 65% RH, and for Viscose Rayon it is 13% under the same conditions.
- Strength: It has varying strengths. Regular rayon loses some of its strength when wet. The highly wet molecules of rayons are strong. The Tensile Strength of the fiber is less when the fiber is wet than when dry. It is 1.5-2.4 gpd in the dry state and 0.7-1.2 gpd in the wet state. For high tenacity variety, the values are 3-4.6 gpd and 1.9 to 3.0 gpd.
- Resiliency: It has low resiliency due to the weak hydrogen bonds. Fabrics wrinkle easily unless given a resin finish.
- Thermo Chemical: Same as that of cotton. If the temperature is too high it scorches and then burns.
- Density: Same as that of cellulose. The density of Viscose rayon is 1.53 g/cc. Rayon filaments are available in three densities: 1.5, 3.0 and 4.5
- Conductivity: It is enough to prevent the buildup of static electric charges.
- Elasticity: The elasticity of Viscose Rayon is less than 2-3%. This is very important in handling viscose yarns during weaving, steering etc when sudden tensions are applied.
- Elongation at Break: Ordinary Viscose rayon has 15-30% elongation at break, while high tenacity rayon has only 9-17% elongation at break.
- The action of Heat and Light: At 300 deg F or more, VR loses its strength and begins to decompose at 350-400 deg F. Prolonged exposure to sunlight also weakens the fiber due to moisture and ultraviolet light of the sunlight.
CHEMICAL PROPERTIES
- Effect of Acids: Hot and concentrated acids cause this fiber to disintegrate.
- Effect of alkali: Strong alkali causes this fiber to swell and lose strength. Weak alkalis do not destroy.
- Miscellaneous: Ultra-violet rays of sun deteriorate this fiber. They are resistant to all insects barring silverfish. Mildew destroys this fiber.
RAYON FIBER CHARACTERISTICS
- Highly absorbent
- Soft and comfortable
- Easy to dye
- Drapes well
The drawing process applied in spinning may be adjusted to produce rayon fibers of extra strength and reduced elongation. Such fibers are designated as high tenacity rayons, which have about twice the strength and two-thirds of the stretch of regular rayon. An intermediate grade, known as medium tenacity rayon, is also made. Its strength and stretch characteristics fall midway between those of high tenacity and regular rayon.
Some Major Rayon Fiber Uses
- Apparel: Accessories, blouses, dresses, jackets, lingerie, linings, millinery, slacks, sports shirts, sportswear, suits, ties, work clothes
- Home Furnishings: Bedspreads, blankets, curtains, draperies, sheets, slipcovers, tablecloths, upholstery
- Industrial Uses: Industrial products, medical surgical products, nonwoven products, tire cord
- Other Uses: Feminine hygiene products
ACETATE RAYON
Today rayon is the generic term for all of the manufactured naturals, while viscose implies a specific process and materials – so viscose is a rayon, but not all
Acetate, acetate rayon, or cellulose is the second oldest manufactured cellulose fibre, with origins dating back to 1865, though it wasn’t until the turn of the century that a process that was commercially satisfactory was developed (interestingly enough, by the Dreyfuss brothers, who were also responsible for the first synthetic indigo dye). Acetate differs from viscose significantly in that acetate is hydrophobic (reacts very poorly to water and must be dry cleaned) and viscose is hydrophilic (loves water and absorbs lots of it). Acetate did poorly at first as a fashion fiber because the initial process was hard to die (as it couldn’t be put in a dye bath) so was used primarily for industry. Acetate also reacts poorly to heat and melts easily – this does mean that it is one of the few semi-natural fibers that can be permanently pressed into pleats. Because of these drawbacks, it wasn’t manufactured commercially on any great scale until at least 1924. Variants of acetate are Triacetate and Diacetate.
The long chain molecule which forms the basis of cellulose acetate is the abundant natural polymers. It is the same basis as is used for viscose and cuproammonium yarns, but in this case of cellulose acetate, most of the Hydroxyl groups have been acetylated.

Acetate Rayon- We know that – Alcohol + Acid –> Ester
If the cellulose is treated with acetic acid under certain conditions the free hydroxyl groups of cellulose are converted into ester groups.
Manufacture of cellulose acetate
Unlike in the case of viscose rayon and cuprammonium rayon, where cellulose is dissolved and regenerated, cellulose acetate is manufactured by converting cellulose into a chemical compound of cellulose ( or chem modified cellulose) which is then dissolved in a suitable solvent ( chloroform or acetone) and spun by evaporating the solvent. Thus while viscose and cuprammonium rayons are regenerated fibers, acetate rayon is regenerated modified fiber.
Raw Material
Cotton
If the degree of substitution is 2.5 to 2.7 then it is known as Diacetate and if the degree of substitute is 2.7 to 2.9 then it is known as triacetate.
The raw material for the acetate rayon is, therefore, the cellulose which comes from either cotton
- Purification of raw material :
- Purification of raw material: Cotton
linters are too short to spun are purchased in form of bales. The bales are broken and thelinters are kier boiled under pressure for a period of 4 to 10 hrs with an alkaline liquor, which may be a solution of either Na2CO3 or NaOH or a mixture of the two. They are then rinsed, washed bleached with sodium hydroxylate, washed and dried. - Pretreatment: The purified cotton
linters are steeped in glacial acetic acid to make it more reactive.
- Acetylating: The pretreated cellulose is transferred and loaded in a closed reaction vessel. A mixture of acetic acid and acetic anhydride in excess is also added to it
.The reaction is hastened by adding a catalyst of sulphuric acid- Acetic Acid – CH3COOH
- Acetic Anhydride – CH3CO —- O —- COCH3
Suitable quantities: - 100 Lbs – purified
linters (Air dry weight) - 300 Lbs – Acetic Anhydride
- 500 Lbs – Glacial acetic acid
The exothermic reaction is so cool to avoid degradation of cellulose. For the first four hours, the temperature is kept at 200C. and then increased to25 to300C for the next 7 to 8 hours.
After 7 to 8 hours the mass becomes gelatinous and very viscous. As a result of this reaction cellulose dissolved in the reaction mixture and it is then considered to be fully acetylated. At this stage, three molecules of acetic acid combine with one molecule of cellulose to form cellulose to acetate. This is known as primary acetate.
Now, this primary acetate is not suitable for the manufacturer of acetate rayon for the reason that it is soluble only in objectionable and expensive solvents such as chloroform. In order to, convert this chloroform i.e. soluble primary cellulose into acetone soluble secondary acetate is known as hydrolysis treatment.
- Hydrolysis: The solution of primary acetate is transferred to another vessel. It is run into the water together with the excess of acetic acid anhydride. The result is a 95% solution of acetic acid in water. The mixture is allowed to stand for 20 hours at a high temperature. During this period Acid hydrolysis of cellulose triacetate takes place. Acetyl groups are replaced by the hydroxyl group. It is an actually partial reversal of acetylating process.
Samples are removed and tested at intervals so that the reaction can be stopped when the acetyl content has been reduced by the desired amount (up to 2.5 molecules of acetic acid per molecule of cellulose). This is now known as secondary acetate which is insoluble in chloroform, but readily soluble in acetone a solvent which is comparatively cheap and not objectionable from the process point of view. - Washing and bleeding: The secondary acetate is though washed, centrifuged and dried at low temperature. In batch production, each batch is tested for viscosity and acetyl content. The blending of different batched is always done to ensure uniformity.
- Dope Preparation: Secondary acetate from various batches is mixed in a closed vessel with the stirrer of about three its own weight of acetone. It will dissolve slowly and completely for about 24 hours thereby giving a preparation known as DOPE.
Well, mixed dopes (Dope contains 25 to 35 % cellulose acetate) from different mixers are again collected and kissed in a blending tank. It is filtered, de-aerated then run into a feed tank. This solution has just about the right viscosity to enable it to be spun into rayon filament.
Acetone being volatile liquid, readily evaporates in warm air, the spinning operation can be very simple. - pinning: The method of spinning adopted is known as dry spinning. It consists of forcing measure amount of the dope solution (cellulose acetate) through spinneret within small chambers or cell through which hot air s fed near the bottom at a temperature of 1000C. This evaporates practically all acetone in dope emerging from the jets.
 Acetone laden air is withdrawn from the top. As the acetone evaporates, solidified filaments of cellulose acetate are left and are collected through an eye. They are wound on the bobbins at the other end of the chamber. Since the cellulose acetate is so well purified before it is dissolved in acetone to form a spinning solution. The resulting rayon filaments are equally pure and require no further treatment except for grading according to quality
Acetone laden air is withdrawn from the top. As the acetone evaporates, solidified filaments of cellulose acetate are left and are collected through an eye. They are wound on the bobbins at the other end of the chamber. Since the cellulose acetate is so well purified before it is dissolved in acetone to form a spinning solution. The resulting rayon filaments are equally pure and require no further treatment except for grading according to quality.Both acetic acid and acetones are sufficiently valuable to the installation of recovery plant. So that there will be the minimum loss of these chemicals. 
Elongation at break is 25% in dry state and 35% in wet state- Acetate Rayon is more sensitive to heat. It begins to weaken at 93 deg C. At 175 deg C it becomes sticky and melts at 260 deg C. Like nylon and polyester it is thermoplastic. Thus permanent crimp, pleats, and creases can be imparted to the garment under carefully controlled conditions.
- 5/20/3s means 55 denier yarn, 20 filaments
and 3 TPI S side. - A moisture content of sec. Cellulose acetate is 6.5% at 70 deg F and 65% RH.
- ( Moisture Content= Wt of water in a material /Total wt of material) ( Moisture Regain= wt of water in a material/ oven dry wt of material)
- ( RH= actual humidity/ humidity of air saturated in water).
- The tenacity of Acetate rayon is 1.4
gpd at the dry state and 0.9gpd at wet state. - Acetate rayon is soluble in acetone, methyl ethyl ketone etc.
- Some degeneration takes place when this fiber is exposed to light but not very serious.
- It is stable to hot water.
- Specific Gravity – 1.32 very similar to that of wool.
- It can also withstand treatment with soap or alkali solution having a pH of not more than 9.5 at temp up to 100 deg C. Therefore it can undergo normal scouring and dyeing operations without affecting the
lustre . - It is unaffected by dilute solutions of weak acids but attacked by strong acids. Concentrated organic acids cause swelling
- It is resistant to attack by bacteria and fungi. Its low moisture content contributes to resistance to mildew.
- It is
non toxic andnon irritating to skin - Only a few striations ( 2-3) are present in the
fibre as can be seen from the longitudinal view. The cross section of the fiber have individual lobes and are round and smooth. It is the smaller number of lobes or serrations of acetatefibres that distinguish thefibre - Chemical Resistance: Cold dilute acid do not affect it, but concentrated acid eg. Acetic acid and formic acids attack it in the cold. Alkalies saponify it i.e. they remove acetyl groups but dilute solutions
upto ph of 9.5 are safe, Marphology : Undermicroscope cross section is lobed.- Resistance
tolight : Some tendering take place when cellulose acetate issxposed tolight but it is usually not very serious a loss of about 15% in tenacity has been reported after 200 hours exposure infade meter. - Biological Resistance: Organisms such as moth and mildew find no nutrient in cellulose acetate cases of damage are extremely rare and are invariably due to the organisms feeding on the oil or finishing material applied to the fiber cellulose acetate will not produce dermatitis.
- Electrical properties: Cellulose acetate is thermoplastic i.e. it softens on heating. It melts about 2300C with decomposition. If a very hot iron is used to iron fabrics made out of it, sticking and eventually fusion occurs.
- Solubility: Readily soluble in some organic solvents eg. Acetone, methyl ethyl ketone, methyl acetate, ethyl lactate and is swollen by a large no. of others including chloroform, methylene chloride,
ethylene chloride etc.
- Purification of raw material: Cotton
BAMBOO RAYONS
Bamboo the plant is wonderfully sustainable; bamboo the fabric isn’t so easy to categorize. There are two ways to process bamboo to make the plant into a fabric: mechanically or chemically. The mechanical way is by crushing the woody parts of the bamboo plant and then use natural enzymes to break the bamboo walls into a mushy mass so that the natural fibers can be mechanically combed out and spun into yarn. This is essentially the same eco-friendly manufacturing process used to produce linen fabric from flax or hemp. Bamboo fabric made from this process is sometimes called bamboo linen. Very little bamboo linen is manufactured for clothing because it is more labor intensive and costly. Chemically manufactured bamboo fiber is a regenerated cellulose fiber similar to rayon or modal. Chemically manufactured bamboo is sometimes called bamboo rayon because of the many similarities in the way it is chemically manufactured and similarities in its feel and hand. Bamboo rayons (from bamboo fibres) – simply viscose or other manufactured naturals made with bamboo as their cellulose base. Bamboo can be processes into a fibre in two ways: mechanically, or using chemicals (as rayon). The mechanical process is expensive and time and labour intensive, and results in a much stiffer, less attractive fabric.
The most common way – and the one widely used for bamboo – is called the viscose process. In this process, cellulose material (such as bamboo) is dissolved in a strong solvent to make a thick, viscous solution that is forced through a spinneret into a quenching solution where strands solidify into fiber. This is sometimes called hydrolysis alkalization or solution spinning because the fiber is “spun” in a chemical solution. The solvent used for this process is carbon disulfide, a toxic chemical that is a known human reproductive hazard. It can endanger factory workers and pollute the environment via air emissions and wastewater. The recovery of this solvent in most viscose factories is around 50%, which means that the other half goes into the environment. Other potentially hazardous chemicals are also used in the viscose process, including sodium hydroxide and sulfuric acid. Because of these environmental issues, Patagonia does not use rayon fabric or bamboo fabric made by the viscose process.The rayon process produces a much nicer fibre, but is not particularly environmentally friendly. In some countries, such as the US, bamboo rayons must be marketed as such unless they are entirely mechanically processed. There are no such rules in New Zealand. It is possible to buy mechanically processed bamboo (it’s rather like a stiff, coarse linen), but its usually blended with other fibres to make it softer and more wearable. I’m reasonably certain that the stripes in my Subtly Striped petticoat are manufactured bamboo.
Alternatives to Rayon Patagonia’s material developers have been investigating bamboo since 2003, but since almost all available bamboo fabric is made using the viscose process, we do not use bamboo fabric in our product line. We are aware of some linen-type bamboo fabric that is processed as bast fiber, but currently we are not using it because we have hemp fabrics that perform well in this type of application. The appeal of bamboo fabric is usually the drape and the hand that is a product of the viscose-type chemical processing. Modern clothing labeled bamboo is usually rayon. The bamboo yarn can also be blended with other textile fibres such as hemp or even spandex. Bamboo rayon fabric is heralded as an amazing product for many obvious reasons. It maintains the same physical properties as cotton, in terms of durability, water absorption, elasticity, etc., but adds wonderful benefits like an extremely soft hand and breathability. Bamboo has often been called the softest sheets in the world, having a 250 thread count sheets being softer than 1000 thread count of cotton. Even a low thread count is a good thread count for bamboo sheets because the molecular makeup of the fibers are softer than cotton could ever be! A good thread count for sheets made from bamboo will always be around 250-300. Even the EPA has noted that “Although the manufacturing process further purifies the cellulose, alters the physical form of the fiber, and modifies the molecular orientation within the fiber and its degree of polymerization, the product is essentially the same chemical as the raw material.”
While specifics can vary, the general process for chemically manufacturing bamboo fiber using hydrolysis alkalization with multi-phase bleaching technology – which is the dominate technology for producing regenerated bamboo fiber – goes like this:
- Bamboo leaves and the soft, inner pith from the hard bamboo trunk are extracted and crushed;
- The crushed bamboo cellulose is soaked in a solution of 15% to 20% sodium hydroxide at a temperature between 20 degrees C to 25 degrees C for one to three hours to form alkali cellulose;
- The bamboo alkali cellulose is then pressed to remove any excess sodium hydroxide solution. The alkali cellulose is crashed by a grinder and left to dry for 24 hours;
- Roughly a third as much carbon disulfide is added to the bamboo alkali cellulose to sulfurize the compound causing it to jell;
- Any remaining carbon disulfide is removed by evaporation due to decompression and cellulose sodium xanthogenate is the result;
- A diluted solution of sodium hydroxide is added to the cellulose sodium xanthogenate dissolving it to create a viscose solution consisting of about 5% sodium hydroxide and 7% to 15% bamboo fiber cellulose.
- The viscose bamboo cellulose is forced through spinneret nozzles into a large container of a diluted sulfuric acid solution which hardens the viscose bamboo cellulose sodium xanthogenate and reconverts it to cellulose bamboo fiber threads which are spun into bamboo fiber yarns to be woven into reconstructed and regenerated bamboo fabric.
- Amine oxides are weak alkalines that act as surfactants and help break down the cellulose structure. Hydrogen peroxide is added as a stabilizer and the solution is forced through spinnerets into a hardening bath which causes the thin streams of viscose bamboo solution to harden into bamboo cellulose fiber threads. The hardening bath is usually a solution of water and methanol, ethanol or a similar alcohol. The regenerated bamboo fiber threads can be spun into bamboo yarn for weaving into fabric.
Other chemical manufacturing processes for bamboo fabric are appearing such as using acetic anhydride and acetic acid with sulfuric acid as a catalyst to form acetate fiber which is then spun into a yarn.
Chemically-manufactured bamboo rayon has some wonderful properties which are adored by conventional and eco-aware designers and consumers:
- Bamboo fabric has a natural sheen and softness that feels and drapes like silk but is less expensive and more durable.
- Bamboo clothing is easy to launder in a clothes washer and dryer.
- Because of the smooth and round structure of its fibers, bamboo clothing is soft and non-irritating, even to sensitive skin. Some people with chemical sensitivities can not tolerate bamboo clothing. We are not sure if this intolerance is due to the intrinsic nature of bamboo but it more likely because of other chemicals added or used during the manufacturing and finishing processes of the clothing.
- Bamboo is naturally anti-bacterial and anti-fungal supposedly because of a bacteriostatis agent unique to bamboo plants called “bamboo kun” which also helps bamboo resist harboring odors. “Kun” is also sometimes spelled “kunh”. The bamboo kun in bamboo fabric stops odor-producing bacteria from growing and spreading in the bamboo cloth allowing bamboo clothing to be more hygienic and to remain fresher smelling.
- Bamboo clothing is hypoallergenic.
- Bamboo is highly absorbent and wicks water away from the body 3 to 4 times faster than cotton. In warm, humid and sweaty weather, bamboo clothing helps keep the wearer drier, cooler and more comfortable and doesn’t stick to the skin.
- The structure of bamboo fibers make bamboo fabrics more breathable and thermal regulating than cotton, hemp, wool or synthetic fabrics.
- Bamboo clothing is naturally more wrinkle-resistant than cotton, and while it might still require ironing after washing, bamboo fabric can be ironed at a lower temperature than cotton. Shrinkage during washing and drying is minimal at warm temperatures.
- Bamboo fibers and fabrics absorb dyes faster and more thoroughly than cotton, modal and viscose with better color clarity. Bamboo fabrics do not need to be mercerized to improve their luster and dye-ability like cotton requires.
Designers such as Kate O’Connor use bamboo fabric as an eco-friendly replacement for silk. Speaking of bamboo eco-fashion, Kate O’Connor calls bamboo fashion “so much cheaper [than silk] and it’s really good for the environment.” “It is the perfect summer fabric” according to Kate O’Connor. Linda Loudermilk, another savvy eco-fashion designer, frequently incorporates bamboo into her eco-fashions. Amanda Shi of Avita has some of the most exciting and originally beautiful eco-fashion in bamboo.
Bamboo the plant and also bamboo the fabric can rate high as an environmentally friendly and renewable resource: - Bamboo grows rapidly and naturally without any pesticides, herbicides or fertilizers.
- Bamboo clothing (both mechanically and chemically manufactured) is 100% biodegradable and can be completely decomposed in the soil by micro-organisms and sunlight without decomposing into any pollutants such as methane gas which is commonly produced as a by-product of decomposition in landfills and dumps.
- Growing bamboo improves soil quality and helps rebuild eroded soil. The extensive root system of bamboo holds soil together, prevents soil erosion, and retains water in the watershed.
- Bamboo grows naturally without the need for agricultural tending and large diesel exhaust-spewing tractors to plant seeds and cultivate the soil.
- Bamboo plantations are large factories for photosynthesis which reduces greenhouse gases. Bamboo plants absorb about 5 times the amount of carbon dioxide (a primary greenhouse gas) and produces about 35% more oxygen than an equivalent stand of trees.
- Bamboo fabrics and clothing can be manufactured and produced without any chemical additives although eco-certification such as Oeko-Tex is necessary to insure that the manufacturing and finishing processes are healthy.
- Currently, there are no known genetically modified organisms (GMO) variants of bamboo. Let’s hope it stays that way.
This gives some feel for how chemically intensive the hydrolysis-alkalization and multiphase bleaching manufacturing processes are for most bamboo fabrics that are promoted as being sustainable and eco-friendly.
LYOCELL / TENCEL
Lyocell is a form of rayon which consists of cellulose
Unlike most rayons, it is strong when wet, and is highly absorbent. It can also be made to mimic a variety of other
Other lyocell processes include Lenzing Lyocell and TITK Alceru. These processes are advantageous because they are environmentally benign, using nontoxic NMMO hydrates instead of toxic carbon disulfide, which can be almost totally recycled. The lyocell fiber has a highly crystalline structure in which crystalline domains are continuously dispersed along the fiber axis. This offers good wet strength as well as excellent dry strength, which makes lyocell water-washable. Further, it shrinks less when wetted by water and dried than other cellulose fibers such as cotton and viscose rayon.
Recently, a new lyocell process, the
The Properties of Lyocell
Comparisons of lyocell with viscose in both laboratory and test markets proved that the
- stronger than any other cellulosic
fibres , especially when wet - easy to process into yarns and fabrics alone or in blends
- easy to blend (unique
fibre presentation) - easy to spin to fine count yarns
- very stable in washing and drying
- thermally stable
- easy to dye to deep vibrant
colours - capable of taking the latest finishing techniques to give unique drape
- comfortable to wear
The Courtaulds’ Lyocell Process
The Courtaulds’ semi-commercial production system is: Dissolving grade

Lyocell Conversion
Lyocell is similar in strength to polyester and stronger than cotton and all other man-made staple
Lyocell shares many properties with other cellulosic
Yarn Manufacture
Tencel can be produced via established yarn manufacturing routes, uses conventional machinery with few major changes to settings or procedures. Its processing performance is influenced by the following properties: • it possesses a non-durable crimp • it has a high modulus • there is a little
As of 2010, Lyocell is more expensive to produce than cotton or viscose rayon. It is used in many everyday fabrics. Staple

MODAL
A generic name for a modified rayon fiber that has high tenacity and high wet modulus. Modal fibers were initially developed in the 1930’s for industrial uses in tires, conveyor belts and hose pipes. Changes in the rayon processing, such as the spin conditions, chemical solutions and stretching sequences, produced rayon fibers with increased crystalline and thus, greater strength. Additional developments in Japan in 1951 by S. Tachikawa lead to the production of rayon with a high wet modulus. These specific types of modal fibers are called polynosic fibers. Modal fibers are dimensionally stable and do not shrink or get pulled out of shape when wet like many rayon. They are also wear resistant and strong while maintaining a soft, silky feel. Modal fibers have found a wide variety of uses in clothing, outwear and household furnishings. They are often blended with cotton, wool or synthetic fibers.
Modal is type of viscose. Modal fabric has softness, good drape and is comfortable for wearing Modal fabric has good moisture regain and air permissibility which is often considered better than cotton fabric, it is a good material for exercise clothing and health suit, which can serve to benefit physiology circulation and health of the body. Modal fabric has level up surface, fine and smooth and velvet, which have the effect of natural silk. The yarn has the character of high strength. The fabric has good softness and brilliant luster. The effect of the finished Modal garment is very good, and the form is steady. The garment maintains anti-crease properties and has relatively easy care.
Modal is a bio-based fiber made by spinning (Spinning is the process of creating yarn (or thread, rope, cable) from various raw fiber materials.)reconstructed cellulose (Cellulose (C6H10O5)n is a long-chain polymeric polysaccharide carbohydrate, of beta-glucose. It forms the primary structural component of green plants. The primary cell wall of green plants is made primarily of cellulose; the secondary wall contains cellulose with variable amounts of lignin. Lignin and cellulose, considered together, are termed lignocellulose, which (as wood) is the most common biopolymer on Earth. Only one group of animals, the tunicates, have evolved the ability to create and use cellulose.)from beech trees. It is about 50% more hygroscopic, or water-absorbent, per unit volume than cotton is. It is designed to dye just like cotton, and is color-fast when washed in warm water. Modal is essentially a variety of rayon.
Textiles made from Modal do not fibrillate, or pill, like cotton does, and are resistant to shrinkage and fading. They are smooth and soft, more so than even mercerized cotton, to the point where mineral deposits from hard water, such as lime, do not stick to the fabric surface. Like pure cotton, modal needs to be ironed after washing.
